Cybersecurity Directive
Medical Devices Regulation
Regulation on Medical Devices for in Vitro Diagnostics
Quality Management System
Environmental Management System
Environmental Management System
Interpretace požadavků MDR, IVDR, GMP a dalších regulací s ohledem na specifika farmaceutického odvětví.
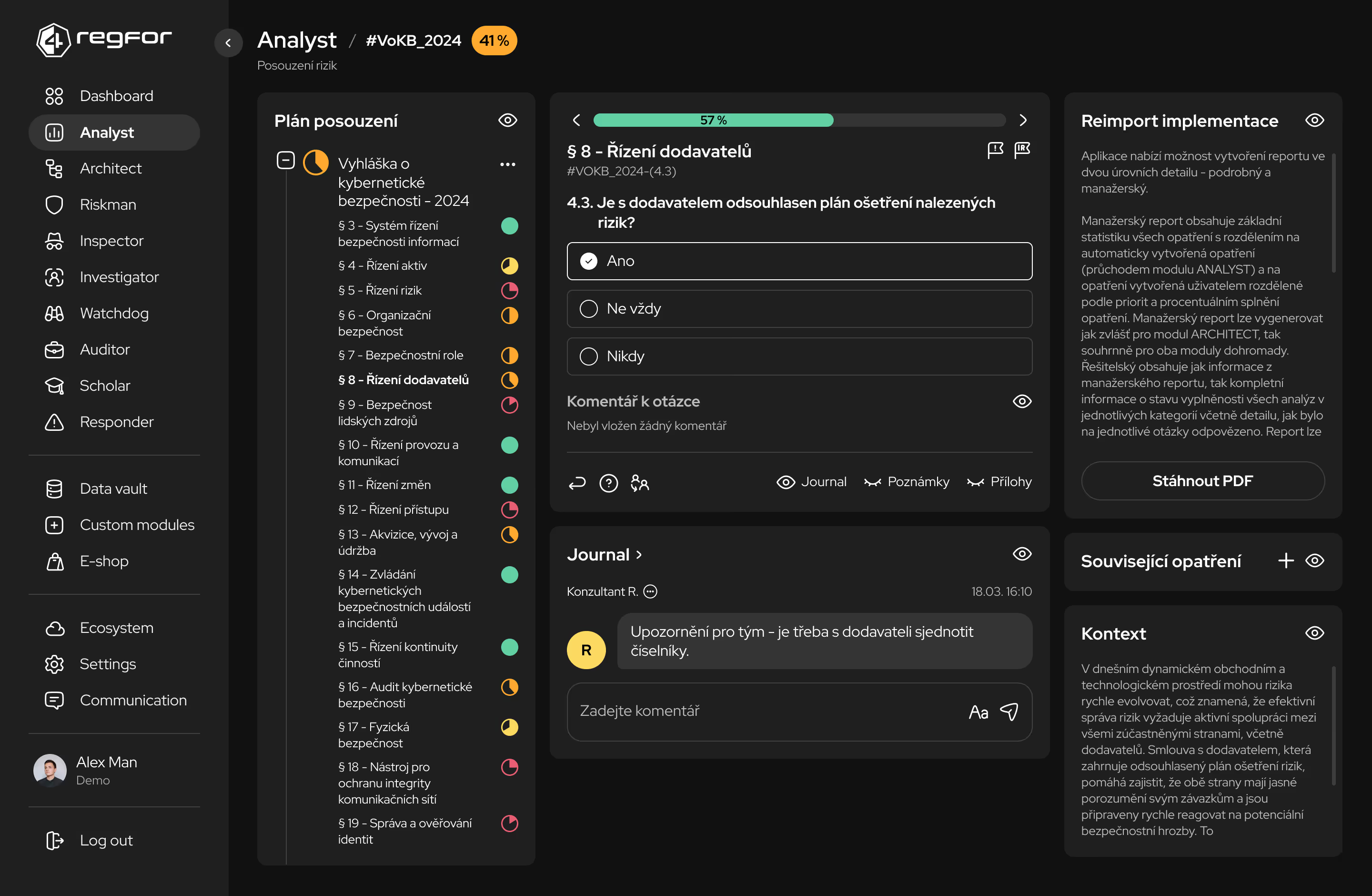
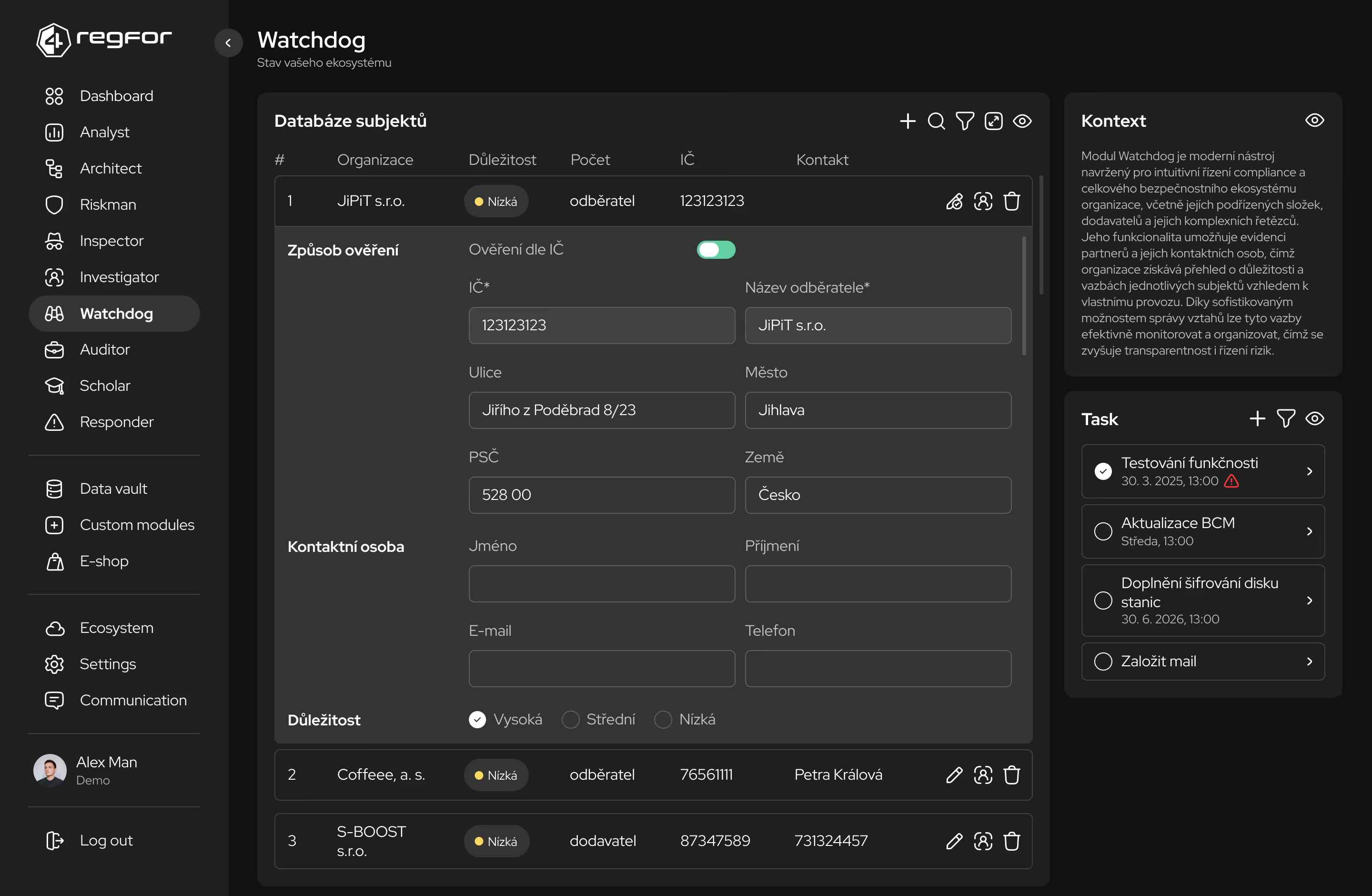
Systematické plánování a sledování implementace opatření podle regulatorních požadavků.
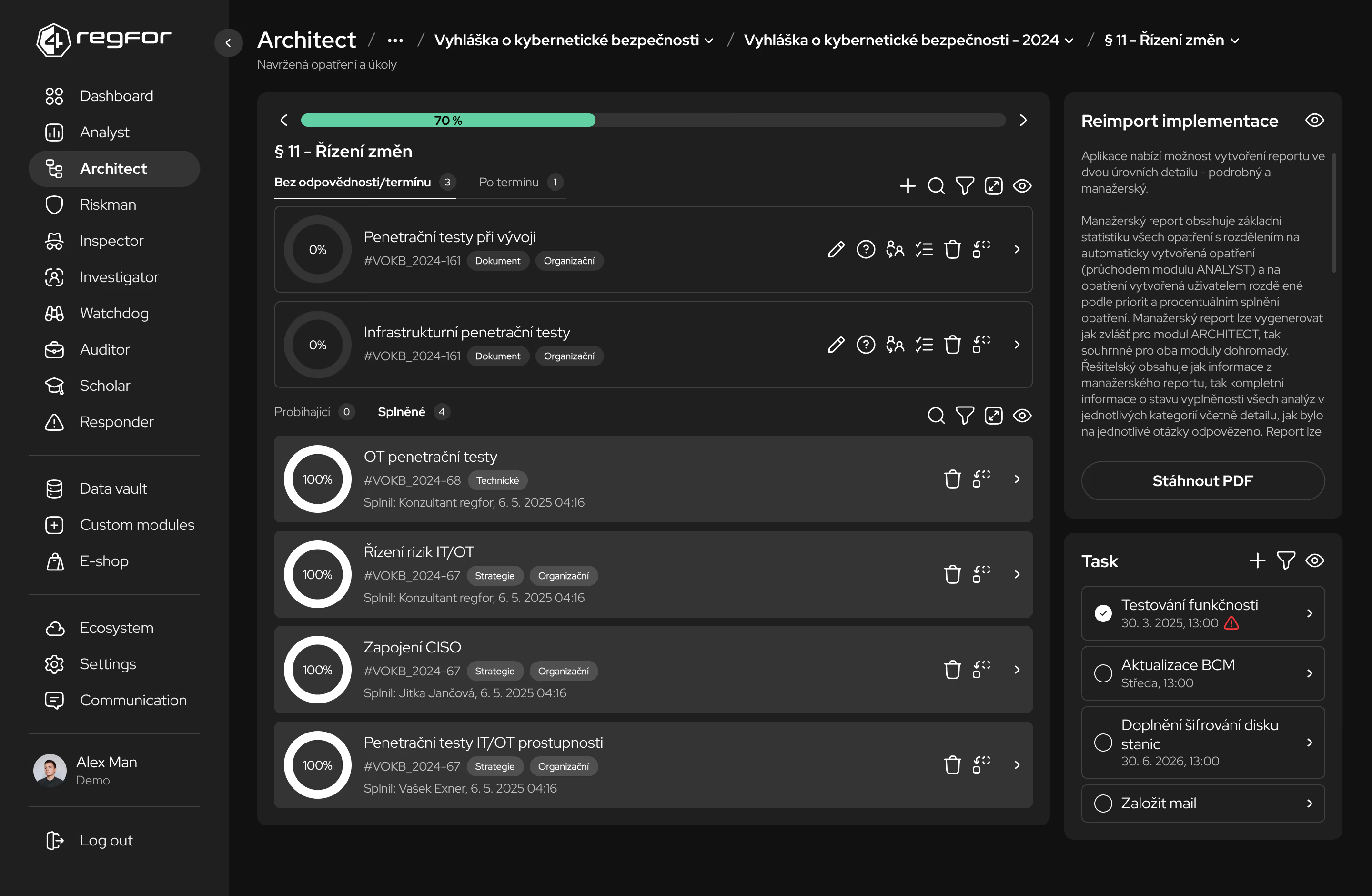
Průběžné kontroly a audity stavu implementace opatření a připravenosti na inspekce.
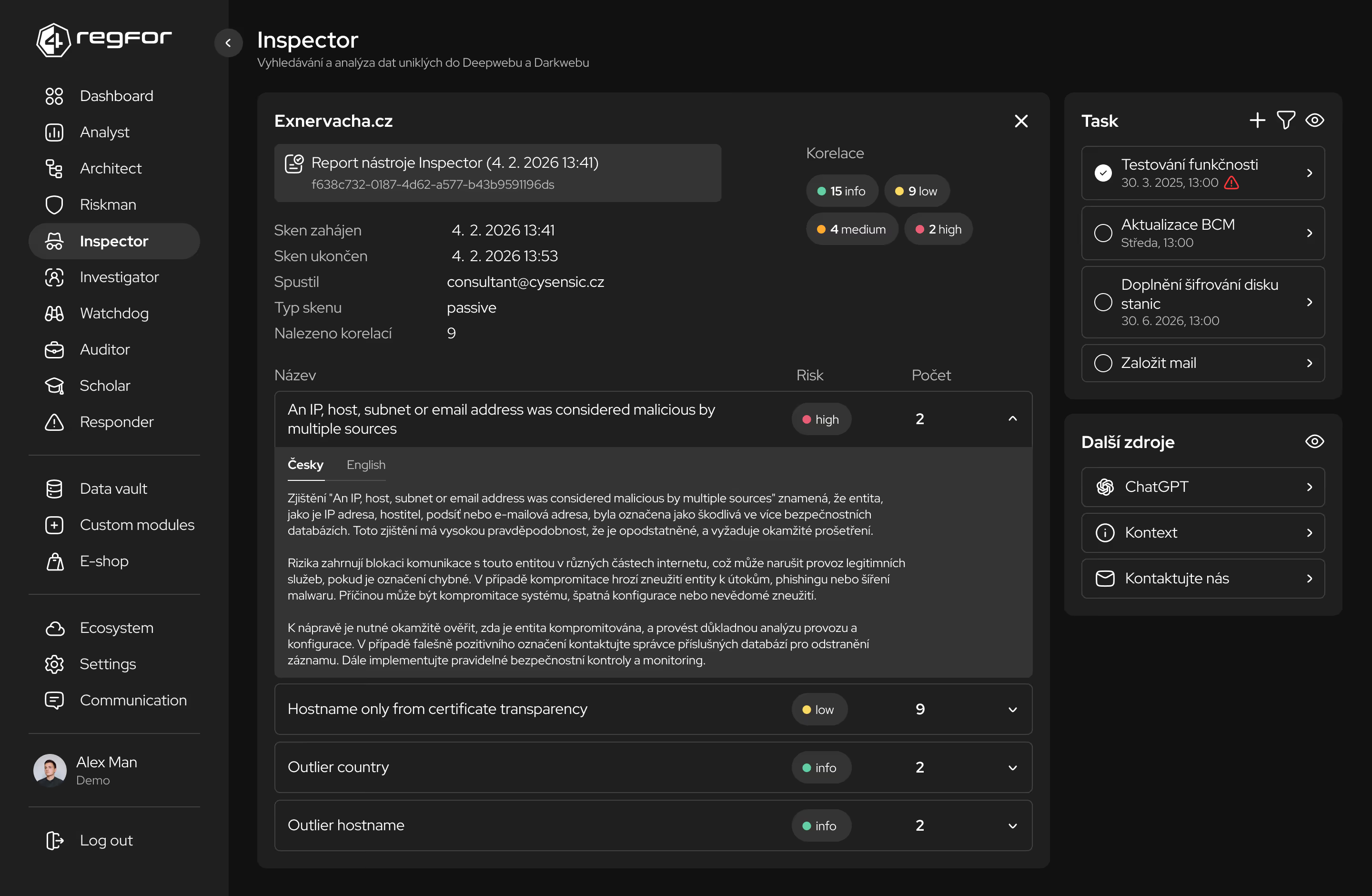
Detailní prověřování dodavatelů s důrazem na řízení rizik dodavatelského řetězce.
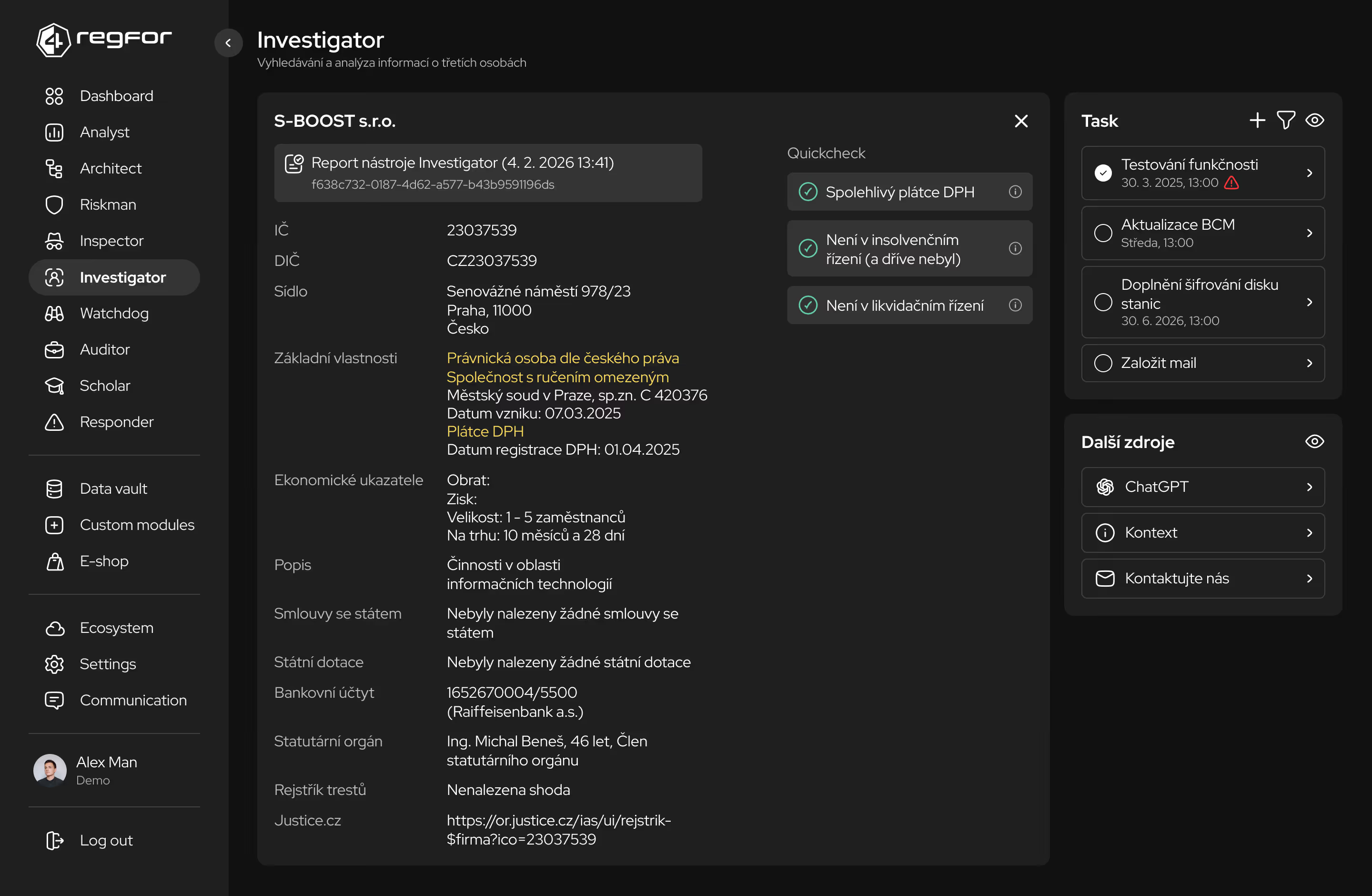
Komplexní řízení rizik včetně hodnocení kritických procesů a závislostí na dodavatelích.
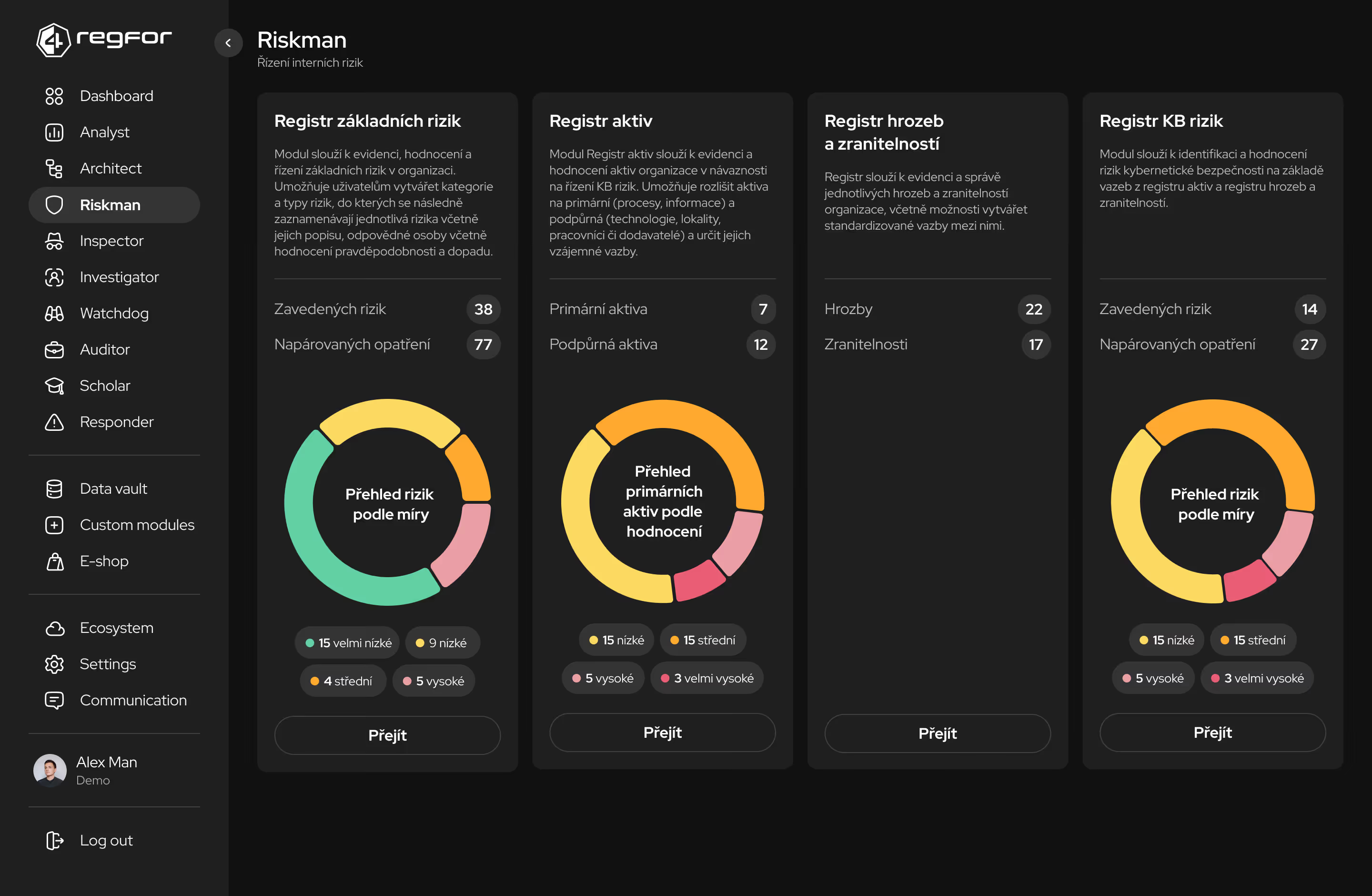
Bezpečné úložiště pro regulatorní dokumentaci a auditovatelné záznamy připravené pro kontroly.
Nepřetržité sledování dodržování požadavků napříč výrobními linkami a laboratořemi.